A pharmaceutical product leaves the manufacturing facility only after passing strict quality checks, regulatory approvals, and packaging inspections. Every batch is verified, sealed, and prepared for distribution through authorized supply chains.
But once the product enters the market, maintaining control becomes more challenging. Pharmaceutical companies must rely on distributors, pharmacies, and healthcare providers to ensure the medicine reaching patients is genuine.
In many markets, counterfeit medicines and unauthorized distribution make this difficult. Packaging may appear authentic, yet visual inspection alone cannot always confirm whether the product is legitimate.
This is why pharmaceutical manufacturers are increasingly adopting phygital packaging solutions. By combining secure physical packaging features with digital verification technologies, phygital packaging enables products to be authenticated instantly through smartphone scanning.
This approach strengthens product security, improves transparency, and helps pharmaceutical companies maintain trust from the factory to the pharmacy.
The Growing Risk of Counterfeit Medicines in Pharmaceutical Supply Chains
Pharmaceutical supply chains often involve multiple intermediaries before a product reaches the patient.
These may include:
- Manufacturers
- Exporters
- Distributors
- Wholesalers
- Pharmacies
Each stage introduces potential risks related to counterfeiting or unauthorized product diversion.
Common threats include:
- Counterfeit medicines replicating genuine packaging using security hologram stickers
- Unauthorized resale through unapproved distributors
- Product diversion into unintended markets
- Repackaging or relabeling of pharmaceutical products
- Difficulty verifying authenticity at the pharmacy level
These risks create serious consequences for pharmaceutical companies.
They can result in:
- Patient safety concerns
- Regulatory compliance challenges
- Distributor conflicts
- Brand reputation damage
- Financial losses from counterfeit circulation
Without reliable authentication systems, confirming product authenticity becomes extremely difficult once medicines leave the manufacturing facility.
Why Traditional Pharmaceutical Packaging Security Is No Longer Enough
Traditional pharmaceutical packaging includes elements such as printed labels, barcodes, batch numbers, and expiry information. These features help identify products and support logistics, but they were not designed to prevent modern counterfeiting.
Today, counterfeiters can replicate packaging designs with increasing accuracy. Even simple security labels can sometimes be visually imitated if they are not supported by authentication systems.
Traditional packaging elements can often be:
- Copied or reproduced
- Reprinted on counterfeit products
- Removed and applied to fake packaging
- Visually replicated using advanced printing technologies
Even standard holographic labels without authentication layers may not provide sufficient protection against modern counterfeiting.
Because of this, appearance alone cannot reliably confirm authenticity.
This is why pharmaceutical companies are moving toward multi-layered packaging security systems that combine physical anti-counterfeit features with digital verification.
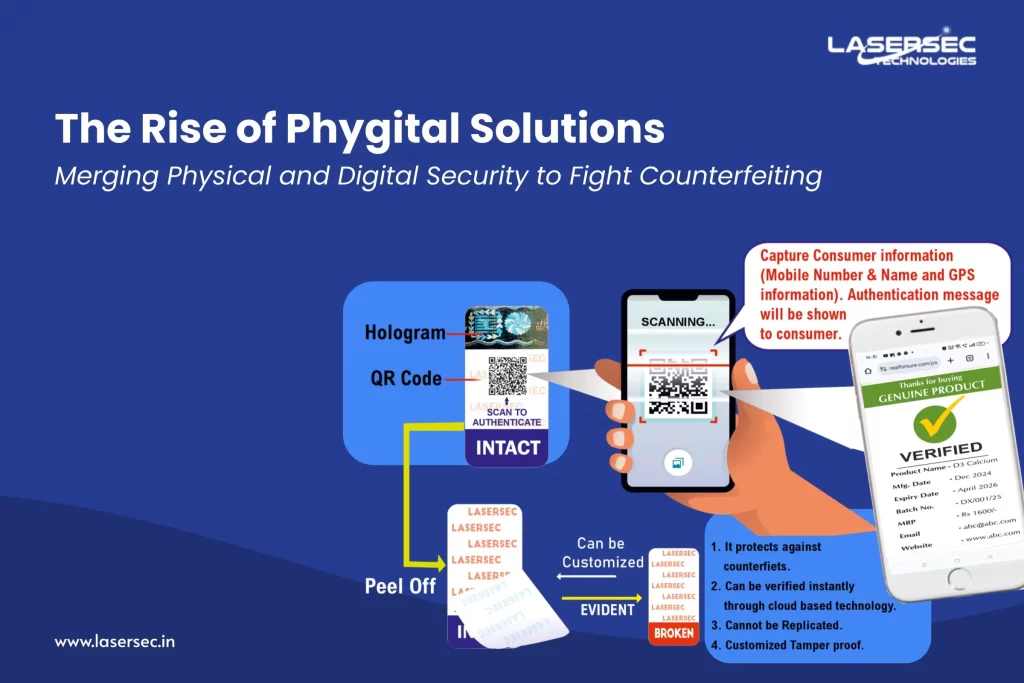
What Is Phygital Packaging in the Pharmaceutical Industry
Phygital packaging integrates physical packaging security with digital authentication systems to create a stronger protection mechanism.
Instead of relying only on visual security features, phygital packaging adds a digital verification layer directly to the product packaging.
In pharmaceutical packaging, this often includes:
- Serialized QR codes
- Unique product identification codes
- Tamper-evident holographic labels like tamper-evident labels
- Secure authentication markers
- Track & Trace verification systems
Each product unit receives a unique digital identity linked to secure manufacturing and distribution data.
When the code is scanned, the authentication system can instantly verify:
- Whether the product is genuine
- Where the product was manufactured
- The production batch details
- The intended distribution market
This transforms pharmaceutical packaging into a secure authentication point for verifying product legitimacy.
How Phygital Packaging Enables Patient-Level Product Verification
One of the most valuable advantages of phygital packaging is that it allows verification directly at the pharmacy or patient level.
When a medicine package includes a secure hologram integrated with a QR code authentication or authentication marker, users can verify the product by scanning the code with a smartphone.
The system checks the unique identifier against secure product records and confirms authenticity instantly.
If suspicious activity occurs, such as duplicate scans or verification from unexpected regions, the system can immediately flag potential counterfeit or diversion risks.
This enables:
- Pharmacists to verify medicines before dispensing
- Patients to confirm product authenticity
- Regulators to investigate suspicious products
- Pharmaceutical companies to monitor verification activity
By enabling verification at the point of use, phygital packaging significantly reduces the chances of counterfeit medicines reaching patients.
Supporting Pharmaceutical Compliance and Regulatory Requirements
Pharmaceutical regulations around the world are increasingly focused on improving traceability and product authentication.
Many markets now require manufacturers to implement systems that support:
- Product serialization
- Track and Trace capability
- Supply chain transparency
- Authentication verification
Phygital packaging helps meet these expectations by integrating physical anti-counterfeit packaging with digital authentication systems such as Track & Trace solutions.
These systems enable regulators and supply chain partners to verify:
- Product origin
- Manufacturing authenticity
- Batch integrity
- Distribution pathways
Authentication capabilities also improve inspection efficiency by providing instant verification during regulatory checks.
For pharmaceutical companies operating across global markets, this transparency strengthens compliance and improves regulatory confidence.
Protecting Pharmaceutical Brand Trust Through Secure Packaging
Trust plays a critical role in the pharmaceutical industry. Patients, healthcare professionals, and regulators must have confidence that medicines are safe and genuine.
When counterfeit medicines appear in the market, they can quickly damage both patient safety and brand credibility.
Phygital packaging helps pharmaceutical companies protect brand trust by enabling reliable product authentication.
Secure holographic packaging such as security holograms combined with digital verification allows genuine products to be distinguished quickly from counterfeit copies.
This capability helps:
- Prevent counterfeit medicines from circulating undetected
- Strengthen trust among healthcare professionals
- Protect pharmaceutical brand reputation
- Ensure patients receive genuine medicines
Over time, authentication systems reinforce long-term trust in pharmaceutical products.
Improving Pharmaceutical Supply Chain Visibility with Phygital Authentication
Pharmaceutical manufacturers often have limited visibility once products leave the factory.
Phygital authentication systems improve supply chain transparency by generating verification records whenever products are scanned using Track & Trace technology.
These records allow pharmaceutical companies to:
- Monitor product movement across regions
- Detect unauthorized distribution channels
- Identify suspicious verification patterns
- Investigate potential counterfeit activity
- Improve supply chain oversight
This visibility allows companies to identify risks earlier and respond quickly before counterfeit products spread widely in the market.
Phygital authentication shifts supply chain protection from reactive response to proactive prevention.
How We Support Secure Phygital Packaging for Pharmaceutical Brands
At Lasersec Technologies, we provide advanced anti-counterfeit packaging solutions designed for pharmaceutical manufacturers operating in complex global markets.
Our phygital packaging solutions combine secure physical packaging with digital authentication technologies, including:
- Security holograms
- Tamper-evident holographic labels
- QR code authentication integration
- Serialized product identifiers
- Track & Trace authentication systems
By integrating these technologies directly into pharmaceutical packaging, we help manufacturers create reliable product authentication systems that work across the entire supply chain.
Our solutions help pharmaceutical companies:
- Prevent counterfeit medicines
- Enable pharmacy-level product authentication
- Strengthen regulatory compliance
- Protect brand reputation
- Improve global supply chain visibility
To explore industry-specific solutions, visit our Pharma anti-counterfeit solutions page.
Strengthening Pharmaceutical Trust from Factory to Pharmacy
Pharmaceutical supply chains continue to grow more complex, and the risks associated with counterfeit medicines remain significant.
Phygital packaging offers a powerful solution by combining secure holographic packaging with digital authentication technologies. This approach enables instant product verification, improves supply chain transparency, and strengthens compliance with evolving regulations.
Most importantly, it helps ensure that genuine medicines reach pharmacies and patients safely.In today’s pharmaceutical industry, phygital packaging is not just a packaging innovation. It has become an essential strategy for protecting patient safety, brand integrity, and long-term trust in pharmaceutical products.